|
Iron Kit (Reg)
how to test |
Iron Kit (Sen)
how to test |
Iron Overload
Hemochromatosis |
|
|
|
Click below for other Metal Test kits |
|
IRON
Number 26 on the "periodic table" of elements
Tests the presence of ions of iron to a high degree of accuracy to
detect contamination in the body and on a wide range of materials in your environment
Iron is the cheapest and one of the most abundant of all metals. The pure metal is not often encountered in
commerce, but is usually alloyed with carbon or other metals. Pure Iron is very reactive chemically, and
rapidly corrodes, especially in moist air or at elevated temperatures. Iron is primarily obtained from the
minerals hematite (Fe2O3) and magnetite (Fe3O4). Huge amounts of iron are used
to make steel, an alloy of iron and carbon.
In addition to helping build the world around us, iron helps keep plants and animals alive. Iron plays a role in
the creation of chlorophyll in plants and is an essential part of hemoglobin, the substance that carries oxygen
within red blood cells. Iron sulfate (FeSO4) is used to treat the blood disease anemia.
Sources
Iron does not occur in nature in useful metallic form. Iron ore is the term applied to a natural iron-bearing mineral in which the content of
iron is sufficient to be commercially usable. Metallic iron, from which steel is derived, must be extracted from iron ore.
Thousands of products having various chemical composition, forms, and sizes are made of iron and steel by casting, forging, and rolling
processes. Good plant sources of iron include dried fruits, whole grains (including wholemeal bread), nuts, green leafy vegetables, seeds and
pulses. Other foods rich in iron but which are usually eaten in smaller amounts include soya flour, parsley, watercress, black molasses and
edible seaweeds. The use of ironware when cooking foods also contributes to dietary intake.
Acquired iron overload occurs when one obtains excessive amounts of iron from repeated exposure through supplementation, diet, iron shots,
blood transfusion. When iron overload is acquired by oral or injected means, the consequences affect the entire body. However, having
prolonged exposure to iron in tobacco directly or indirectly (second hand tobacco smoke) or iron-containing asbestos products can result in
iron loaded lung cells.
Up to 22% of the iron in meat is absorbed, while only 1-8% is absorbed from eggs and plant foods. If the body stores fall, the rate of iron
absorption rises. About 40% of the iron in animal foods is in a form called haem iron, while the remainder, and all the iron in plant foods,
is in the less well absorbed non-haem form. Iron absorption can also be reduced by tannins (e.g. in tea) and phytates (found in nuts, grain
and seeds).
The absorption of iron from plant foods is improved by the presence in a meal of vitamin C (ascorbic acid), other organic acids such as
malic acid (e.g. in pumpkins, plums and apples) and citric acid (in citrus fruits).
Risk groups of iron overload include:
- Those who use tobacco products on a regular basis or who are exposed to tobacco smoke.
- Those exposed to iron-containing asbestos, patients with thalassemia, sideroblastic anemia or other conditions requiring blood transfusion to remedy anemia.
- Anyone taking excessive amounts of iron; or receiving iron shots.
- One who consumes on a regular basis a limited diet of red meat, tobacco products and alcohol.
Toxicity
Chronic iron (Fe) overload is characterized by increased focal or generalized deposition within the tissues. On tissue examination, this has
been commonly termed hemosiderosis. When excess Irondeposition is associated with tissue injury or the total body Iron estimate is >5 gm, the
term hemochromatosis has been applied
Individuals with hemochromatosis absorb too much iron. Iron cannot be excreted therefore the excess builds to toxic levels in tissues of
major organs such as the liver, heart, pituitary, thyroid, pancreas, lungs, and synovium (joints). These overburdened organs cease to
function properly and eventually become diseased.
Toxicity Symptoms
Hemochromatosis can develop into diseases such as diabetes, heart trouble, arthritis, liver disease, neurological problems, depression,
impotence, infertility, and cancer.
Symptoms of acquired iron overload might include:
- chronic fatigue
- arthritic pain in joints
- loss of libido (sex drive) or impotence
- amenorrhea (premature cessation of menstrual cycle)
- changes in skin color such as jaundice, bronze or gray-olive colored skin, a tan without being in the sun, redness in the palms of the hands
- abdominal pain
- weight loss
- shortness of breath
- chest pain
- heart arrhythmia
- depression
- elevated blood sugar
- hypothyroidism
- enlargement of spleen
- elevated liver enzymes (ALT/AST)
Excess iron lowers the immune system. Many diseases will show a poor outcome unless any excess iron is removed: AIDs, cancer and hepatitis,
for example.
Check out the iron in your body with our easy to use, home-based, HMT Iron Regular Test Kit
or HMT Iron Sensitive Test kit
 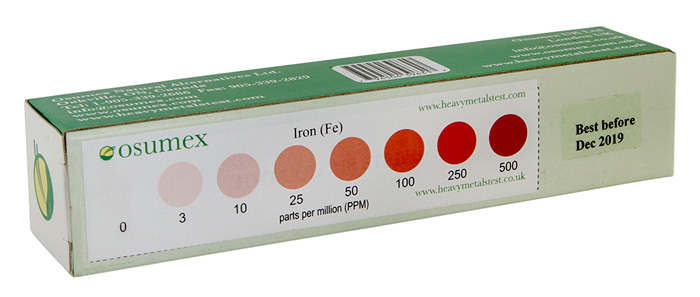
HMT Iron Regular Test kit with color strip for results analysis
 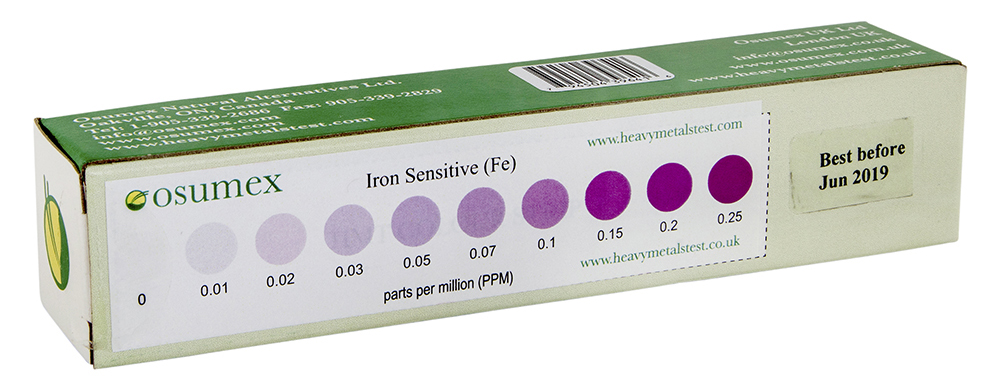
HMT Iron Sensitive Test kit with color strip for results analysis
Osumex HM-Chelat is most effective in eliminating heavy metals contamination in the body

The above information is provided for general
educational purposes only. It is not intended to replace competent
health care advice received from a knowledgeable healthcare professional.
You are urged to seek healthcare advice for the treatment of any
illness or disease.
The Food Standard Agency UK has not evaluated these
statements. This product is not intended to diagnose, treat, cure, or prevent
any disease.
|


